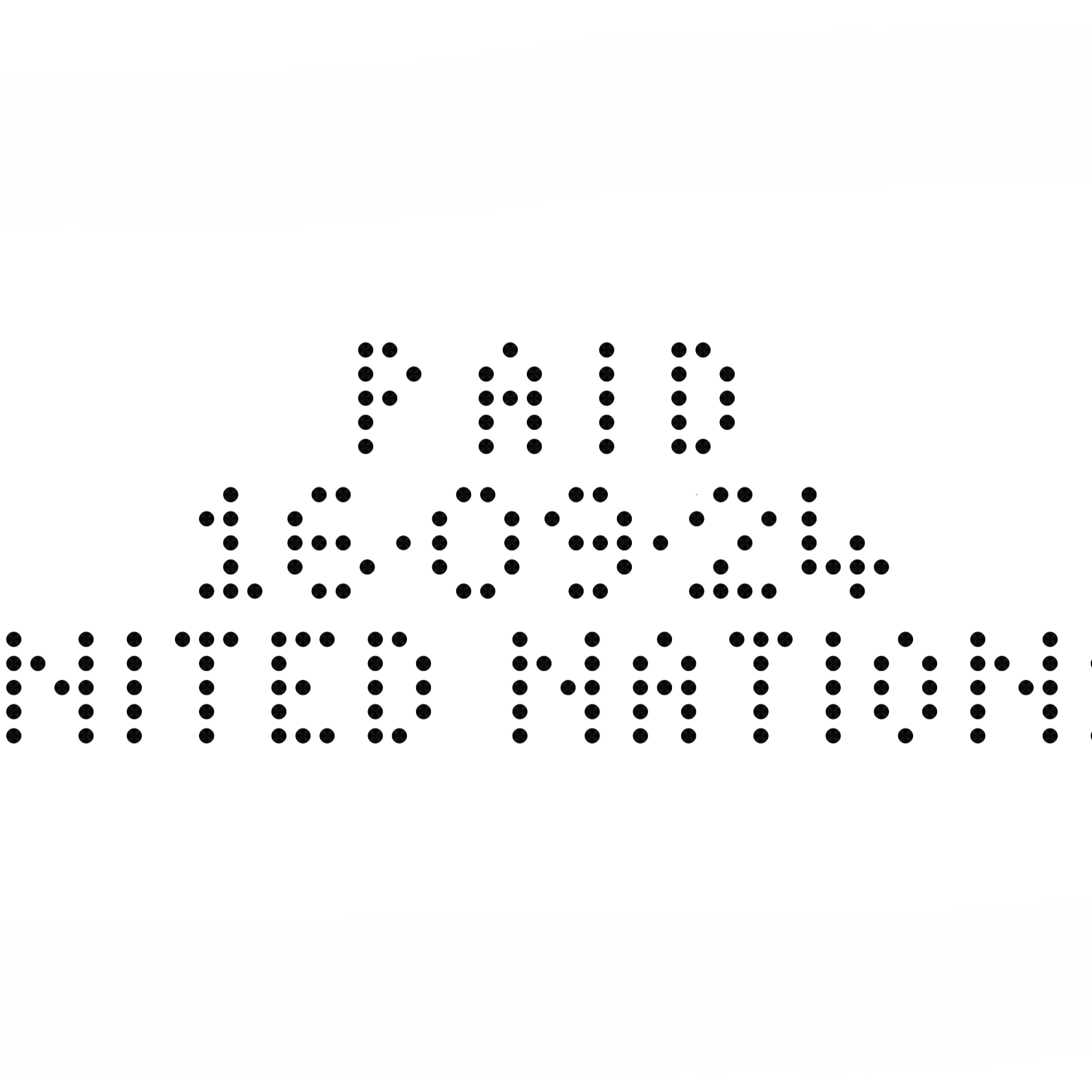In highly regulated pharmaceutical environments, every stage of the process is designed to ensure control, traceability, compliance, and document security.
Yet, there is one step that is often underestimated: how information is marked on documents and packaging.
Expiration dates, batch numbers, and production codes: seemingly simple elements, yet they represent a critical point in quality management.
The hidden challenge
Many traditional marking systems introduce variability:
- readability that can degrade over time
- risk of alteration or counterfeiting
- dependence on external factors (inks, environmental conditions)
- misalignments in manual processes
In regulated environments, even minor inconsistencies can lead to non-conformities, rework, or critical findings during audits.
Regulations and traceability requirements in the pharmaceutical sector
In the pharmaceutical sector, marking is not just a simple operational step.
It is a control point.
International regulations, such as Good Manufacturing Practices (GMP) and data integrity guidelines (ALCOA+), require that all critical information be:
- accurate
- durable in readability over time
- non-alterable
- traceable throughout the entire product lifecycle
Towards a new standard
For this reason, an increasing number of organizations are adopting approaches based on physical, permanent markings integrated directly into the material.
A marking that is not “applied,” but embossed.
In this context, systems such as Perfodoc are positioned, designed for environments where precision and reliability are non-negotiable.
Thanks to dry marking technology, the machine enables direct embossing on documents, labels, and packaging:
- Expiration dates
- batch numbers
- production codes
The result is a permanent, legible, and tamper-proof marking, aligned with the traceability requirements of the pharmaceutical sector.
Process integration
Its robust structure, positioning accuracy, and ability to process multiple sheets simultaneously make the Perfodoc/D suitable for:
- production documentation (batch record)
- quality records
- packaging and labels
- certifications and official documents
All while ensuring consistency, repeatability, and control, even in high-intensity operational environments.
Would you like more information?
If you’d like to explore how to integrate a manual dry marking system into your production or receive tailored advice, feel free to contact us with no obligation.
Request a free quote or more information: we’ll help you identify the solution that best fits your needs.